Wastewater treatment processes often require an additive for pH control to neutralize acid wastewater. Magnesium hydroxide can be an excellent cost efficient alternative to caustic soda (sodium hydroxide) for pH control in aerobic and anaerobic biological wastewater treatment plants.
Magnesium hydroxide as substitute for caustic soda in pH neutralization
Both magnesium hydroxide and caustic soda are used to increase alkalinity or neutralize acids in industrial processes. Caustic soda is a strong alkali that rapidly raises to pH 12 or higher. Magnesium hydroxide on the other hand reacts more slowly and increases the pH up to 9 or 10. Since magnesium hydroxide buffers at a lower pH, it is safer for microorganisms in biological treatment plants. Magnesium hydroxide is non-hazardous and therefore safer to handle than caustic soda.
When it comes to dosing volumes, magnesium hydroxide dosage can be reduced by 25% while maintaining the same alkalinity compared to a 50% caustic soda solution. Reason being that magnesium hydroxide contains more hydroxide ions per molecule than caustic soda. Taking into account the price difference, total savings can be up to 60%. A distinguishment to take into consideration is that magnesium hydroxide reacts more slowly than caustic soda.
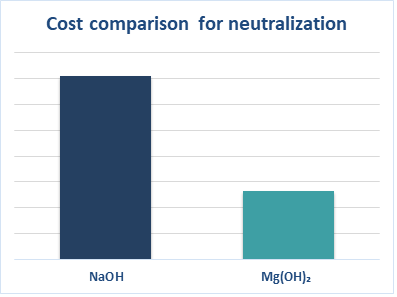
Figure 1. Cost comparison of neutralization based on 600 dry metric ton NaOH consumption.
Applications
Magnesium hydroxide is used for effluent treatment in aerobic and anaerobic biological wastewater treatment plants in industrial and communal wastewater plants. It is also applied in metal precipitation or odor control. In flue gas treatment, magnesium hydroxide is used to remove sulphur oxide compounds and pH adjustment.
Meet magnesium hydroxide: the safe alkali and a cost efficient substitute for caustic soda in pH neutralization.
