pH control is a critical step in many industrial processes where acidic liquid streams need to be neutralized. Stable pH levels are essential to protect equipment and environment, to ensure process efficiency and maintain consistent operating conditions. Magnesium hydroxide offers a safe and controlled solution for pH neutralization, combining high alkalinity with a natural buffering effect. This prevents extreme pH fluctuations and supports a stable, reliable process performance across a wide range of applications.
Safe and controlled pH adjustment with magnesium hydroxide
pH control is essential in industrial processes where acidic liquid streams need to be neutralized. Stable pH levels protect equipment, ensure process efficiency and support compliance with environmental standards.
Magnesium hydroxide offers a safe and cost-efficient solution for pH neutralization. Its controlled reaction and natural buffering capacity prevent extreme pH peaks and ensure stable process conditions.
Magnesium hydroxide vs caustic soda
Magnesium hydroxide is a strong alternative to caustic soda for pH correction. While caustic soda rapidly increases pH to high levels, magnesium hydroxide reacts more gradually and buffers at pH 9 to 10.
This reduces the risk of overdosing and makes it safer for sensitive processes. In addition, magnesium hydroxide contains more hydroxide ions per molecule, resulting in lower consumption and improved cost efficiency.
Key benefits:
- Controlled pH adjustment
- Lower chemical consumption
- Safer handling
- Stable process conditions
Use case: industrial wastewater pre-treatment
An industrial production site in the UK has been using magnesium hydroxide for pH neutralization of acidic wastewater streams for several years, replacing caustic soda as the standard alkali.
The process requires stable pH conditions prior to biological treatment. Magnesium hydroxide is dosed to neutralize the acidic stream before it enters the aeration tanks. Its controlled reaction and buffering capacity ensure a stable pH, preventing harmful fluctuations during aeration.
After neutralization, the water is aerated to remove organic compounds and subsequently discharged to a municipal treatment plant for further treatment.
Result: stable pH control, reliable biological performance and a safer, more controlled alternative to caustic soda.
Applications
Magnesium hydroxide is used for direct pH control in liquid streams across industries such as:
- Chemical processing and process water neutralization
- Metal and surface treatment (acid bath neutralization)
- Food and beverage processing
- Wastewater treatment

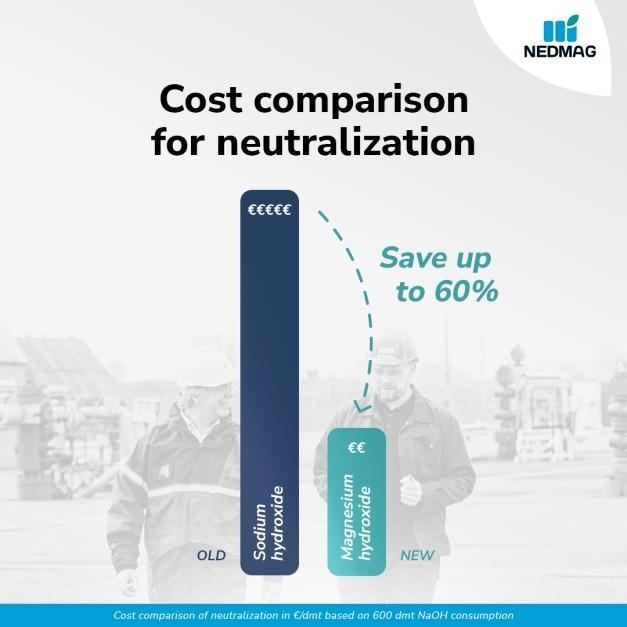
Cost efficient neutralization
Magnesium hydroxide offers a clear cost advantage over traditional alkalis such as caustic soda. Thanks to its higher alkalinity, less product is required to achieve the same neutralization effect. In practice, this can reduce dosing volumes by up to 25 percent.
Combined with improved process stability and safer handling, this results in lower total operating costs and fewer process disruptions. Rather than correcting overdosing or pH spikes, operations remain stable and predictable.


